Study: No Evidence the HPV Vaccine Prevents Cervical Cancer – 54% Increase in Cervical Cancer in British 25 to 29-Year-Olds
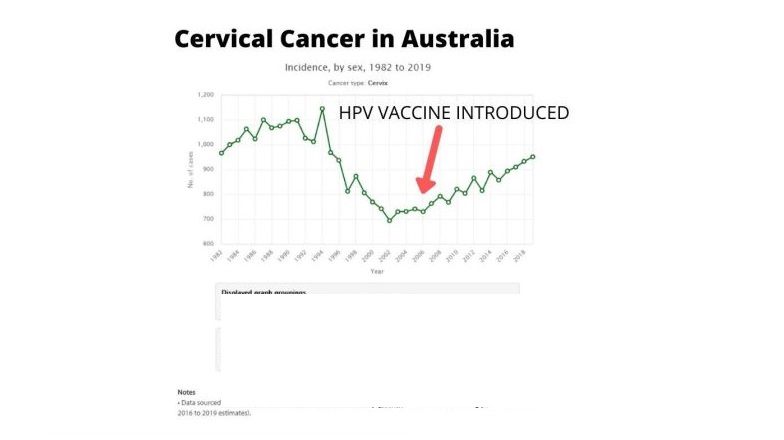
Human papillomavirus (HPV) vaccines hit the global marketplace in the mid-2000s. From the start, public health agencies enthusiastically promoted HPV vaccination as the “best way to protect [young people] against certain types of cancer later in life.” However, a blistering new study by British researchers—and new data showing that cervical cancer rates are surging in British 25- to 29-year-olds—raise numerous questions about officials’ inflated claims. The study’s results indicate, instead, that the jury is still out on whether HPV vaccination is effective. The question is far from academic because, prior to Britain’s introduction of HPV vaccination in 2008, cervical cancer rates had been trending sharply downward. In fact, between the late 1980s and mid-2000s, cervical cancer rates halved. Now, Britain’s leading cancer research charity (Cancer Research UK) reports a steep 54% rise in cervical cancer in one of the very age groups that first received the vaccine.